
This discrepancy challenges medical device companies, as they must struggle to meet multiple standards or lose access to international markets.  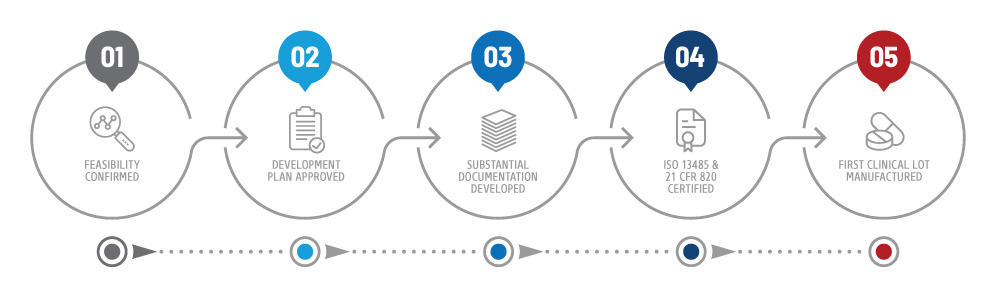
Food and Drug Administration (FDA) has considered their Quality System Regulation (QSR) to be the minimum criteria for medical device manufacturing since 1996, international standards differ. For decades, there has been a divide in the medical device regulatory landscape.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
